Jiangxi Xintao Technology Co., Ltd.
There is chemical industry there is Xintao chemical packing.
The adsorption separation of carbon molecular sieves is to use the selective adsorption separation and purifying gas or liquid mixture of porous solid substances. A complete adsorption separation process is usually composed of adsorption and absorbing (undertaking) cycle operation. The carbon molecular sieve process is divided into transformer adsorption and temperature change adsorption. The temperature change adsorption is to complete the circulation operation by adjusting the temperature. The transformer is taken through the physical adsorption process. As a adsorbent, carbon molecules are separated by transformer adsorption methods to separate nitrogen and oxygen in the air to reach the method of purifying nitrogen.
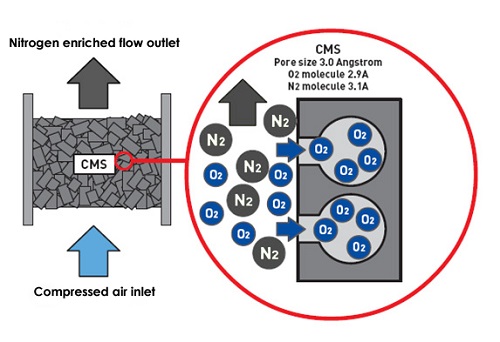
N2 and O2 in the separation of carbon molecular sieve in the air are based on the difference between the two in the diffusion rate. Both N2 and O2 are non -polar molecules, and the molecular diameter is very close (O2 is 0.29nm and N2 is 0.31nm. Because the physical properties of the two are similar, the binding force of the carbon molecular sieve is not much different. From the perspective of thermodynamics (absorbing balance) The adsorption of carbon molecular sieve on N2 and O2 is not selective, and it is difficult to separate the two.
However, from the perspective of dynamics, because the carbon molecular sieve is a-type rate separation adsorption agent, N2 and O2 are in the micropores of carbon molecular sieves in micropores There are obvious differences in the diffusion speed. For example, when: 35'C, the diffusion speed of O2 is 2.0x100, and the speed of O2 is 30 times faster than N2. Therefore ! (Separate in it, so that the N2 in the air can be purified.
Because the adsorption separation process is a process of rate control, the control of the adsorption time (that is, the control of the adsorption-relief loop rate) is important. When the amount of adsorption agent, adsorption pressure, and gas flow rate are certain, the suitable adsorption time can be determined by determining the penetrating curve of the attached column.

E-mail: export@xt988.com
Copyright © Jiangxi Xintao Technology Co., Ltd. | All Rights Reserved
We are here to help you! If you close the chatbox, you will automatically receive a responsefrom us via email. Please be sure to leave yourcontact details so that we can better assist